 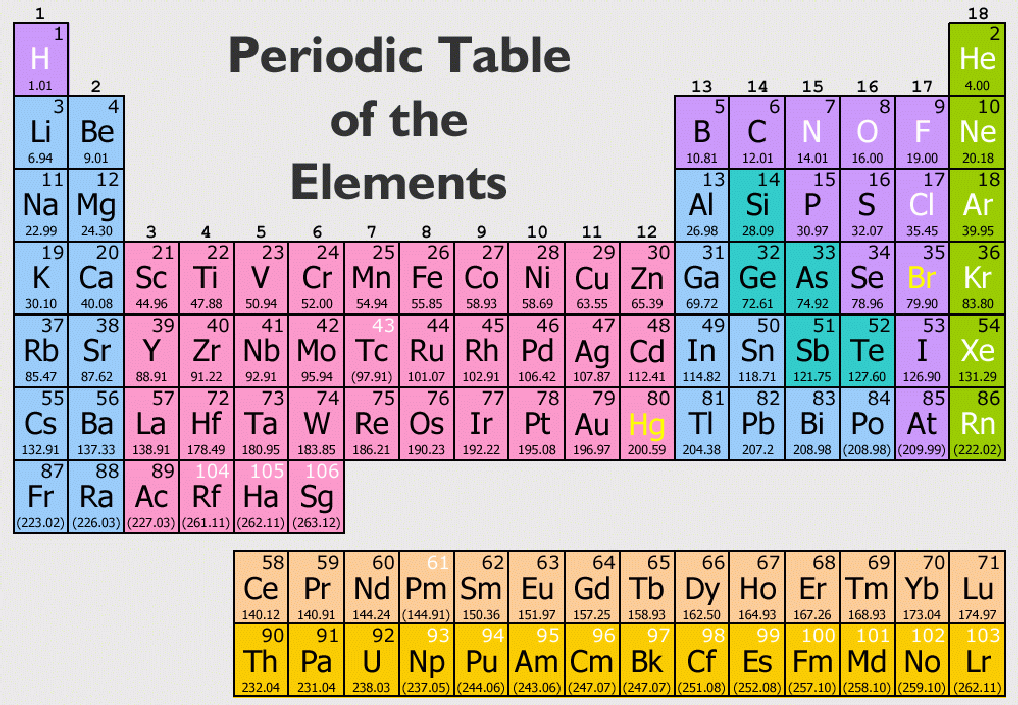
The atomic symbols are hence the easiest tools to quickly obtain the essential information about an element. Read more about atomic structures and isotopes While calculating the number of electrons in an ion, we should subtract the positive charge (for cation) from the number of protons and add the negative charge (for anion) to the number of protons. Carbon – 14, on the other hand, has 8 neutrons.īoth isotopes of chlorine contain an equal number of protons and electrons. The carbon-12 has 6 protons, six neutrons, and 6 electrons since the number of protons & electrons must be equal for it to be neutral. The number of protons, neutrons, and electrons can be easily obtained by the following method.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
